Title
Orthotopic T cell receptor replacement by advanced non-viral cell engineering
Research Area
Hematology, Oncology, Clinical Immunology, Microbiology, Immunology
Project Summary
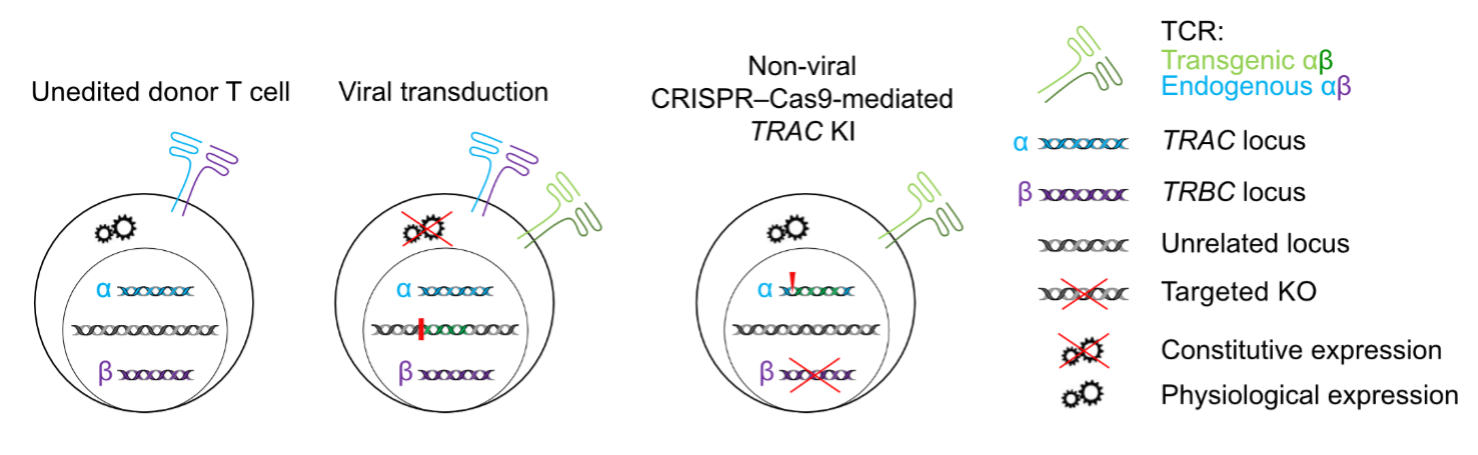
During the last funding period we have developed and further characterized in-depth CRISPR/Cas9-guided orthotopic T cell receptor (TCR) replacement (OTR). We demonstrated that OTR results in engineered T cells with physiological TCR expression, predictable functionality and largely improved epitope-specific cytotoxicity. TCRs recognize a plethora of unique antigens and associated epitopes. Besides necessary reactivity to self-epitopes, which provides important survival signals and may influence tissue tropism to some extent, a substantial fraction of encountered foreign or neo-epitopes can be recognized by individual TCRs. Thus, every TCR harbors a unique (cross-)reactivity profile. Natural T cell responses are polyclonal, entailing many different TCRs even within defined antigen-reactive T cell populations. Out of these, few TCRs with particular characteristics (e.g. high affinity for a target epitope, cross-reactivity for epitope-escape variants, multi-epitope target cell reactivities) seem to be particularly important for immediate or long-term immune protection.