Title
Extending and controlling lymphocyte functionalities by engineering cytokines and their receptors
Research Area
Biochemistry, Cell Biology, Immunology
Project Summary
Lymphocytes critically depend on cytokines to regulate and control their diverse immune functions. Cytokines allow lymphocytes to be activated when needed, remain active even under adverse conditions and control their activation to maintain immune homeostasis and protect the host. At the same time, several cytokines can suppress desired immune cell functions in the tumor microenvironment, a major challenge in adoptive immunotherapy. Cytokine and receptor engineering thus provide many opportunities for current key challenges in adoptive immunotherapy. Building on our work and collaborations from the first funding period, this project will continue to seize these opportunities by using rational cytokine and receptor engineering.
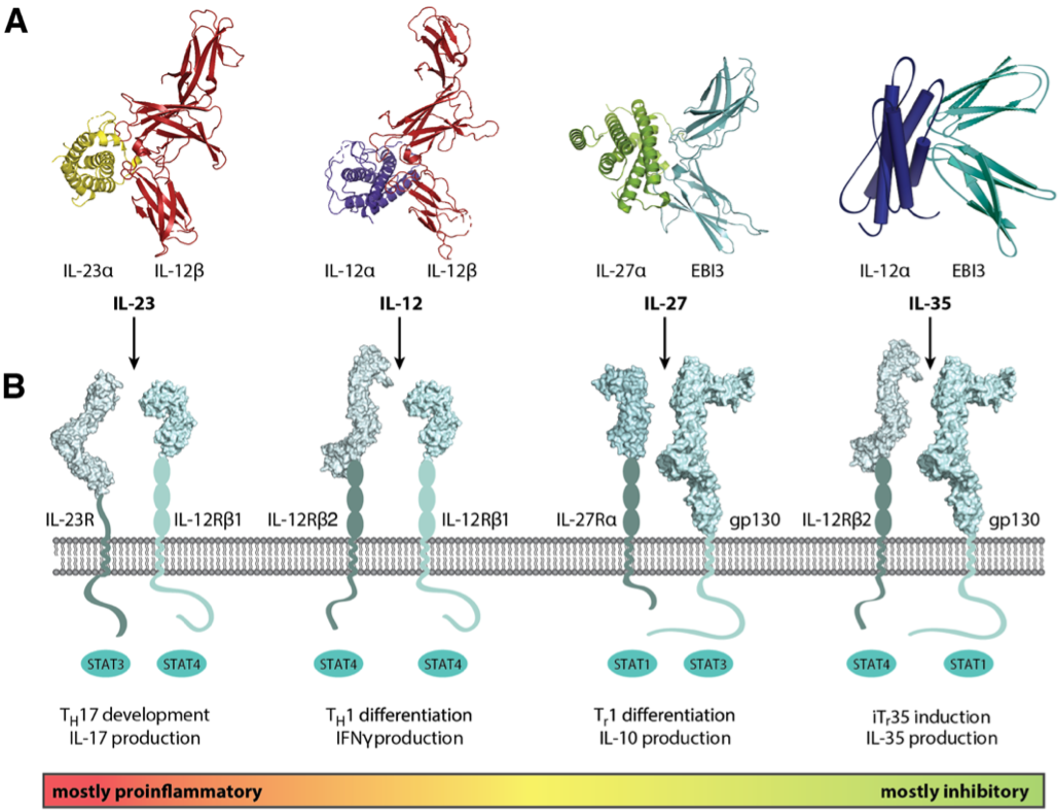
Local restriction of immune function is currently a major limitation for T cell therapies against solid tumors and also limits antiviral therapies. Equipping engineered lymphocytes with suitable immune-activating cytokines is a promising avenue to overcome this limitation and to maintain immune cell functionality even in adverse immunosuppressive microenvironments. Interleukin 12 (IL-12) has very desirable properties to (re-)activate immune responses in immunosuppressive environments, but toxicity currently limits its transfer into the clinics. The first major research goal of our proposal is thus to develop IL-12 variants that increase the potency of adoptive immunotherapy while at the same time being well-tolerable. In close collaboration with several other groups of this consortium, our goal is to bring engineered immune cells equipped with such IL-12 variants into the clinics.
The second major research goal of this project is to engineer and validate bio-orthogonal cytokine:receptor systems that do not occur in humans and thus can be used without interfering with natural signaling pathways. This will add new ways of controlling the functions of engineered lymphocytes, which is important for optimizing functionality but also for maintaining safety. The third goal of our proposal is to develop new tools to re-program the cytokine milieu and/or its functions in the direct neighborhood of engineered lymphocytes. We will do this by inactivating or re-purposing cytokine functions in the proximity of engineered cells. Specifically, we will develop novel approaches to overcome IL-23-mediated promotion of cancer development and growth by engineering IL-23 receptors as cytokine sinks. Taken together, our project will continue to leverage rational cytokine and receptor engineering to overcome major current challenges in adoptive immunotherapy. This will only be possible in close collaboration with groups of complementary expertise in this consortium.