Project Summary
For pediatric acute leukemia (AL) it is a novel concept that the immune system may play a dual role – contributing to both anti-leukemic immunity and possibly also to disease progression. In this project, we start with clinical data from a bedside-to-bench approach analyzing the local interaction between AL and the immune system and apply these data to overcome disease-promoting mechanisms through lymphocyte engineering. We have identified bone marrow T cells (bmT), functioning like tumor-infiltrating lymphocytes (TILs), as key immune components capable of either controlling or facilitating leukemia. Importantly, AL establishes a microenvironment that promotes leukemic cell survival and impairs T cell function and may enable immune evasion. This immunosuppressive milieu also limits the efficacy of immune-based therapies such as chimeric antigen receptor (CAR-) T cells. In the bedside-to-bench translational part, we performed multi-OMICS analyses (transcriptomics, epigenomics, and immune phenotyping) on bmT from several cohorts of children with AL.
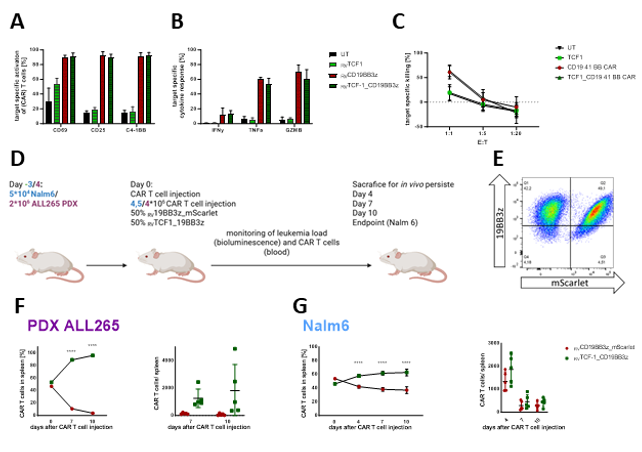
These studies uncovered relevance of bmT with the inhibitory receptor TIM-3 (HAVCR2) emerging as a central marker of T cell dysfunction and relapse risk. Building on this insight, we established a virus-free CRISPR/Cas9-based knock-in system to integrate anti-CD19 CAR constructs into respective genes, offering a robust and controlled CAR expression platform. Besides CAR knock-in into the T cell receptor alpha chain (TRAC) gene, we developed a novel strategy to link transgene expression to the endogenous HAVCR2 (TIM-3) promoter, thereby transforming an inhibitory signal into a context-dependent activation/effector mechanism. Proof-of-concept experiments demonstrated that this approach enables effective anti-leukemic CAR responses in vitro and in vivo, supporting the concept that activation signals can be regulated by endogenous inhibitory gene promoters. Furthermore, we showed in the first funding period that transgenic expression of TCF1, a key regulator of memory and stem-like T cell states, is reduced in bmT and overexpression leads to improved early CAR-T cell expansion and in vivo persistence. This provides a powerful tool to enhance T cell fitness and durability in hostile microenvironments. The overarching goal of this project is to develop combinatorial T cell engineering strategies that overcome the specific mechanisms of AL-induced T cell dysfunction. Work in the future funding period will include mechanistic work-up of bmT dysfunction in patient samples, site-specific CAR integration via CRISPR/Cas9-mediated homology-directed repair (HDR), context-dependent activation via inhibitory receptor gene promoters, and transcription factor modulation such as TCF1 and FOXO1 to support a T cell fate towards long lasting responses. By targeting both, functional exhaustion and limited persistence, this approach seeks to enhance the efficacy, safety, and durability of CAR-T cell immunotherapy in pediatric AL. This includes promoter-specific expression control, minimized off-target effects, and precise genome integration, representing an approach towards a next-generation GMP-platform for engineered cell therapies.
More