Title
Evaluating the therapeutic potential of genetically engineered CAR-Tregs for the treatment of inflammatory bowel disease
Research Area
Immunology, Genetics, Genetic Engineering, Treg Biology
Project Summary
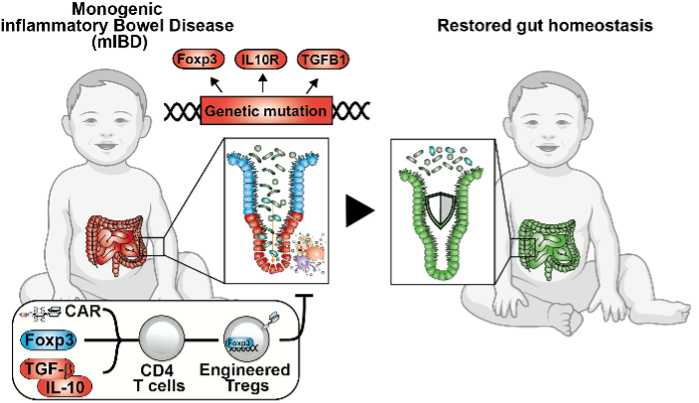
IBD is a chronic or relapsing inflammatory condition of the gastrointestinal tract that has emerged as a global disease burden. Children with very early-onset IBD (VEO-IBD), particularly those with monogenic IBD (mIBD), often suffer from severe, therapy-refractory disease, highlighting the need for novel therapeutic strategies. Studies on monogenic forms of VEO-IBD have provided critical insights into key regulators of intestinal immunity and inflammation. Besides genetic aberrations, disease pathogenesis is driven by a combination of immune dysfunctions, dysbiosis of the intestinal microbiota, environmental factors, and compromised intestinal epithelial barrier function. Quantitative and qualitative defects of regulatory T (Treg) cells disrupt immune homeostasis and may contribute to intestinal inflammation and autoimmunity. Preclinical studies have highlighted the therapeutic potential of adoptive Treg transfer in inflammatory disease models, yet a clinically optimized Treg-based therapy specifically designed for chronic colitis remains not available.
In the proposed project, we aim to evaluate the potential of genetically engineered antigen-specific Tregs with chimeric antigen receptors (CAR-Tregs) for the treatment of monogenic and common inflammatory bowel disease (IBD) (Fig. 1) In Aim 1, we will engineer gut-specific CAR-Tregs capable of overexpressing key immunoregulatory cytokines (e.g., IL-10, TGF-β1) and/or the master transcription factor Fopx3 by gene editing and/or modulation via microbial metabolites to enhance their potency for targeted treatment of mIBD or autoimmune enteropathies caused by Foxp3, IL-10 and TGF-β1 deficiencies. The potency of CAR-Tregs will be assessed ex vivo by examining the antigen-specific activation and suppression of effector T cell responses as well as elucidating epithelial barrier integrity and proinflammatory responses in immune-intestinal organoid co-culture systems. In Aim 2, we will evaluate the therapeutic potential and lineage stability of gut-specific CAR-Tregs in acute and chronic colitis models (e.g., DSS and TNBS) of Il10 and Tgfb1 knockout mice by assessing the progression of colitis through clinical activity scores, histology, as well as immune cell and transcriptional profiling. In Aim 3, we will evaluate the generalizability of our approach for patients with common IBD by determining the therapeutic efficacy of optimized gut-specific CAR-Tregs in preclinical humanized mouse models of colitis.
In summary, our study will provide critical insights into the therapeutic potential of CAR-Tregs for children with severe mIBD, advancing precision medicine. Using monogenic conditions as a proof-of-principle, we aim to establish a therapeutic framework that broadens the applicability of CAR-Tregs to common IBD and other autoimmune or autoinflammatory disorders.