Title
Adapter CAR technologies to steer T cell function and track T cell fait through in vivo imaging with radioligands
Research Area
CAR-T cell engineering, molecular imaging, theranostics
Project Summary
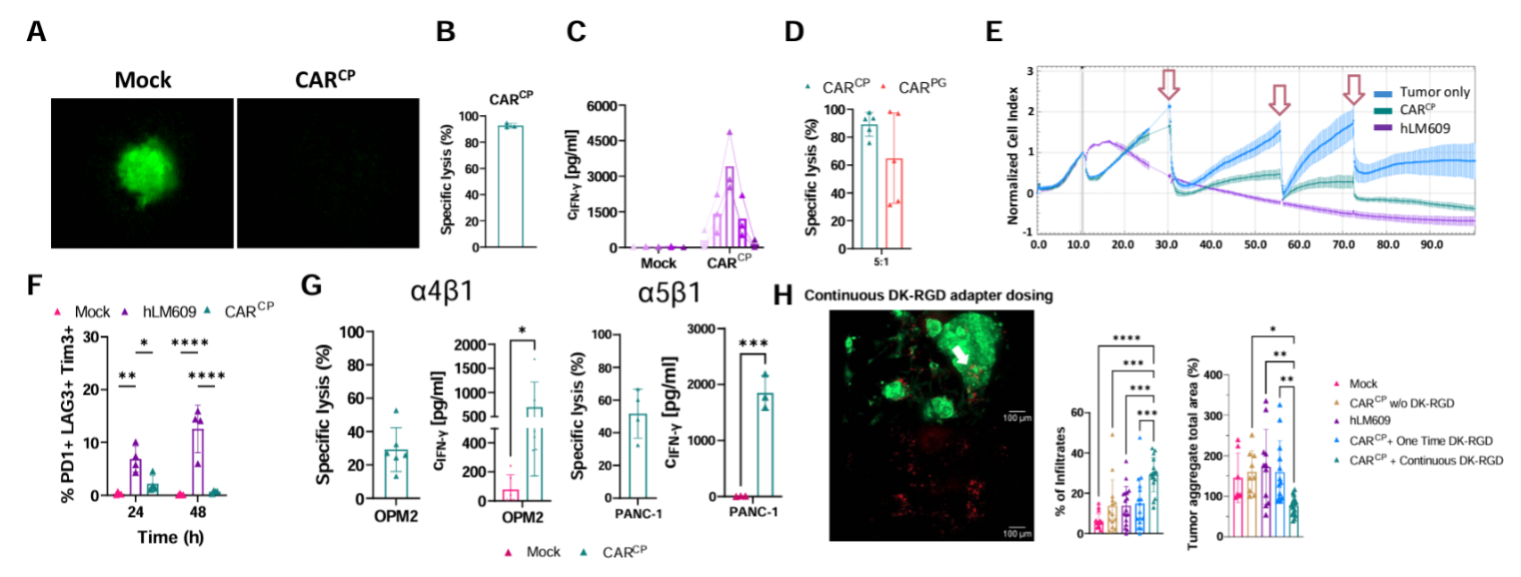
This project is dedicated to the investigation of programmable adapter CARs, that are conceptually attractive because one or several tumor antigens can be targeted concurrently or sequentially to increase or maintain therapeutic pressure against difficult-to-treat cancers. We are pursuing a novel platform that is based on diketone (DK)-peptidomimetic adapters that covalently bind to a reactive lysine residue in the scFv of a chemically reactive CAR (CARCP, clone h38C2). In the first project period, we have demonstrated that T cells expressing this CARCP can be programmed with RGD-DK peptidomimetic adapters to confer specific and potent anti-tumor reactivity against αvβ3-integrin expressing cancers (incl. melanoma, breast, lung and renal cancer) in preclinical models in vitro and in vivo. A major challenge and knowledge gap for the use of CARCP-T cells is how to achieve and maintain adapter molecule concentrations in tumor tissue that ensure optimal CAR-T cell function. Both underdosing and overdosing of adapters – relative to CAR-T cells and tumor cells – will lead to ineffective therapy. To address this challenge, we propose in vivo imaging to determine the concentration of CAR-T cells, CARCP and tumor antigen by PET. This approach is based on our prior work with RGD-based radioligands for αvβ3-integrins that are already commonly used for PET imaging in animal models and patients. Further, we have recently developed a highly sensitive reporter gene approach for imaging CAR-T cells in vivo (DTPA-R). Using these tools, the strategic objective for this project in the second funding period is to enhance the therapeutic aspect of tumor targeting with the CARCP adapter platform and to integrate the diagnostic aspect of CARCP-T cell tracking and monitoring in vivo. The specific aims are to derive detailed insights into the pharmacokinetic (PK) of radiolabeled RGD-DK adapters and CAR-T cells through in vivo PET imaging and mathematical modelling in order to devise refined adapter designs and dose schedules that are optimal for anti-tumor efficacy.
We will also investigate a new class of adapter molecules based on peptides that contain metal complexes of the chelator DOTA. To target these molecules, we will use a scFv with picomolar affinity for DOTA-metal complexes to construct a CAR. The key advantage of this approach is that many DOTA-labeled compounds are already used clinically in nuclear medicine for imaging and treating various cancers. These ligands are metabolically stable in humans and have optimal PK attributes for tumor uptake. Thus, DOTA-binding CAR-T cells (CARDOTA) have enormous potential for clinical translation. In the work program for the second funding period, we will compare the anti-tumor reactivity that can be conferred with the CARCP and CARDOTA approach and the ability to support in vivo imaging with radiolabeled adapters. Furthermore, we will determine if the DTPA-R system can be used to read out the functional state of CAR-T cells in vivo. For this aim we will express DTPA-R under the control of inducible promoters or alternatively study bivalent ligands that bind to the reporter protein and a T cell activation marker (e.g. CD69).
The overall ambition of this project is to develop CAR-T cells that can be programmed to confer specific anti-tumor efficacy and that can be tracked and monitored through PET imaging in vivo. These advances will also be useful for TCR-transgenic T cells in infectious and malignant diseases and contribute to the overall objectives of the CRC.