Title
Developing polyclonal TCR-T cell therapies using CRISPR base and prime editing
Research Area
Immunology, Cell Engineering
Project Summary
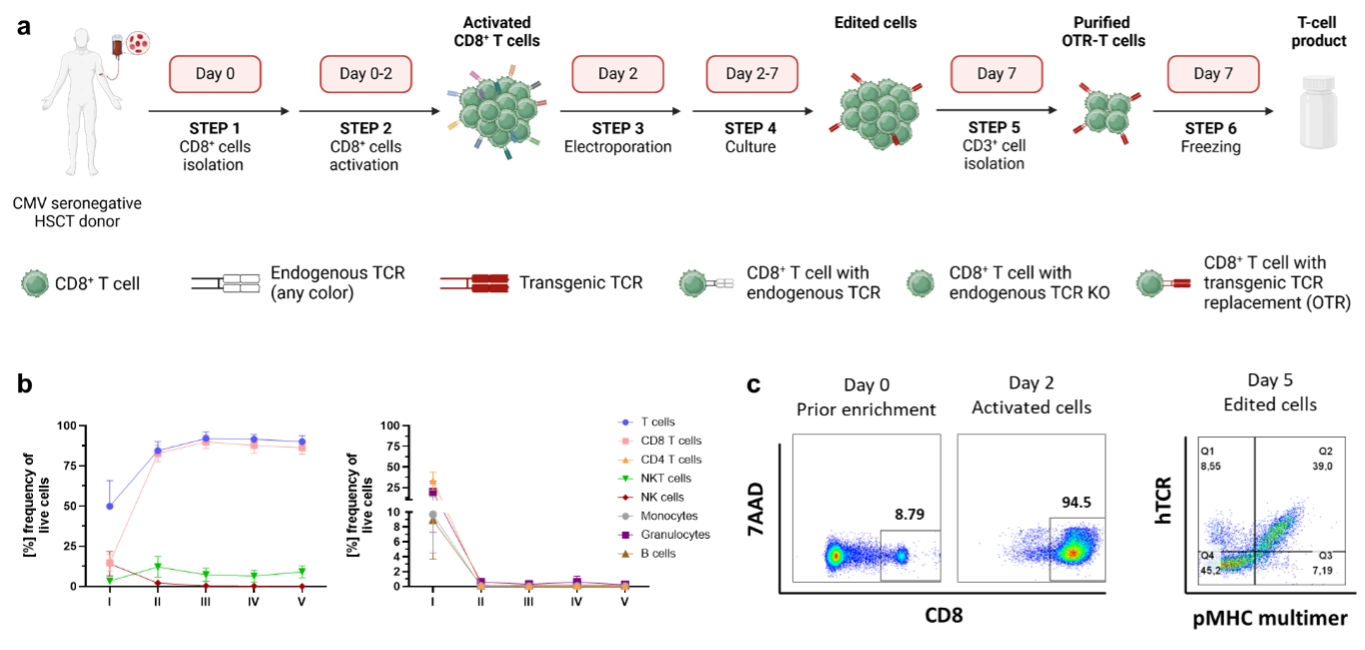
Adoptive T cell therapies with polyclonal, naturally occurring multi-specific T cells have led to clinical remissions in patients with viral reactivations, including Cytomegalovirus (CMV), and cancer. However, the ability to predict the in vivo functionality of these products remains limited. Reprogramming T cells using transgenic receptors enables the generation of cell products with well-defined efficiency and specificity. The method for transgenic receptor integration can affect the quality and homogeneity of the generated cell product. CRISPR/Cas9 can site-specifically knock-in transgenic T cell receptors (TCRs) at the TRAC or TRBC locus – a technique referred to as orthotopic TCR replacement (OTR). OTR places the transgenic TCR under the control of the endogenous TCR promoter, enabling physiologically regulated TCR expression and homogenous cell products. Conventional CRISPR/Cas9 can induce undesired editing outcomes, including chromosomal translocations and truncations, compromising the safety of the T cell product and potentially complicating clinical translation. Advanced CRISPR editing platforms, like base (BE) and prime editing (PE), exhibit a reduced rate of undesired editing outcomes, representing promising technologies for advancing T cell immunotherapy.
The overall goal of this proposal is to generate OTR TCR-T cells using advanced genetic engineering with BE and PE, enabling the development of multifunctional TCR-T cell therapies that combine the advantages of physiological polyclonal T cell products with the predictability of engineered monoclonal T cells. To do this, we will 1) optimize the targeted transfer of transgenic TCRs through BE and PE, 2) perform an extensive functional and phenotypical benchmarking of BE/PE-edited versus Cas9-edited OTR-T cells, 3) assess the biological relevance of gene editing-induced translocations and chromosomal truncations by following their clonal dynamics over time, and 4) establish OTR at the TRBC locus for the monoallelic integration of transgenic TCRs, thus allowing the pooled generation of polyclonal T cell products.