Title
Engineering CAR-NK cells for sustained tissue functionality
Research Area
Medicine, Hematology, Oncology, Clinical Immunology, Microbiology, Immunology
Project Summary
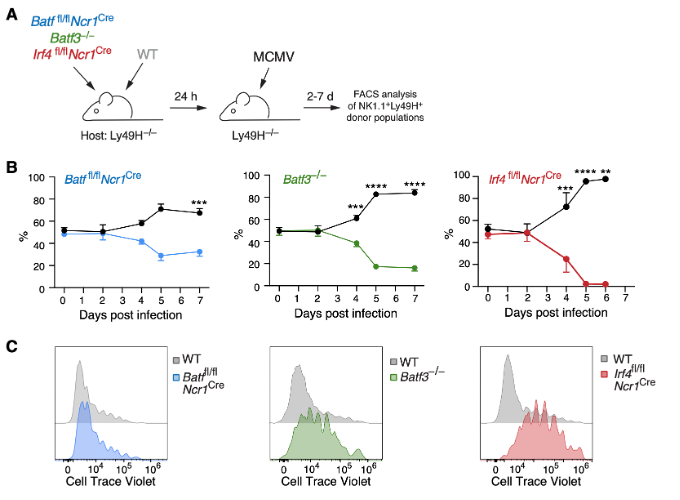
Chimeric Antigen Receptor Natural Killer (CAR-NK) cells represent a promising alternative to CAR-T cell therapy, potentially offering several advantages including a superior safety profile, off-the-shelf availability, reduced risk of graft-versus-host disease (GvHD), broad targeting capabilities, and versatile mechanisms of action. Despite these benefits, a significant challenge remains the ability of both CAR-NK and -T cells to provide sustained therapeutic responses and effectively home to and eliminate solid tumors or metastatic lesions within tissues. We have significantly contributed to our understanding of the development and transcriptional regulation of innate lymphoid cells (ILCs) and NK cells in tissues during development. Our recent work has further revealed the intriguing differentiation potential of circulating conventional NK cells into tissue-resident effector cells during ontogeny and in response to infection. Notably, we have identified a novel population of CD69hi TCF1hi tissue-resident NK (trNK) cells, which exhibits remarkable similarities to CD56bright TCF1hi NK cells that are enriched in various human tissues. Our findings suggest that a balanced expression of the transcription factors Tcf1, Hobit, and Blimp1 plays a critical role in regulating the establishment of trNK cells and their subsequent acquisition of effector functions and cytotoxicity. Furthermore, we have discovered that AP1/ATF-family transcription factors BATF, BATF3 and their potential interacting partner IRF4 are pivotal for the efficient in vivo expansion of NK cells during infection.
Building on these insights, our project aims to delineate the transcriptional requirements of CAR-NK cells (murine and human) within the context of adoptive cell therapy. To enhance the in vivo expansion potential and functionality of CAR-NK cells, we will employ in ‘Aim 1’ a range of genetic engineering techniques including overexpression, knockdown, and CRISPR-mediated targeting of aforementioned transcription factors. In ‘Aim 2’, we will explore possibilities to engineer CAR-NK cells that retain”stemness” and the capacity to differentiate into TCF1hi tissue-resident NK cells and provide effector function in suppressive tissue and tumor microenvironments. In ‘Aim 3’, we will build on our previous observations that NK cell effector function and target cell recognition can be modulated through CD4+ T cells. Therefore, we hypothesize that providing CD4+ T cell help to CAR-NK cells will improve CAR-NK cell expansion and target cell interaction. To test this hypothesis, we will closely collaborate with B06 (Kastenmüller) and evaluate a novel strategy for engineering of CD4-help for CAR-NK cells.
By integrating our current understanding of trNK cell biology with advanced genetic engineering approaches, we want to overcome existing limitations in CAR-NK cell therapy – 1) pronounced and sustained in vivo expansion, 2) differentiation and functionality in tissues, and 3) protective effector function. This research has the potential to significantly advance the field of cancer immunotherapy by providing innovative strategies for enhancing the efficacy and durability of CAR-NK cell responses.