Project Summary
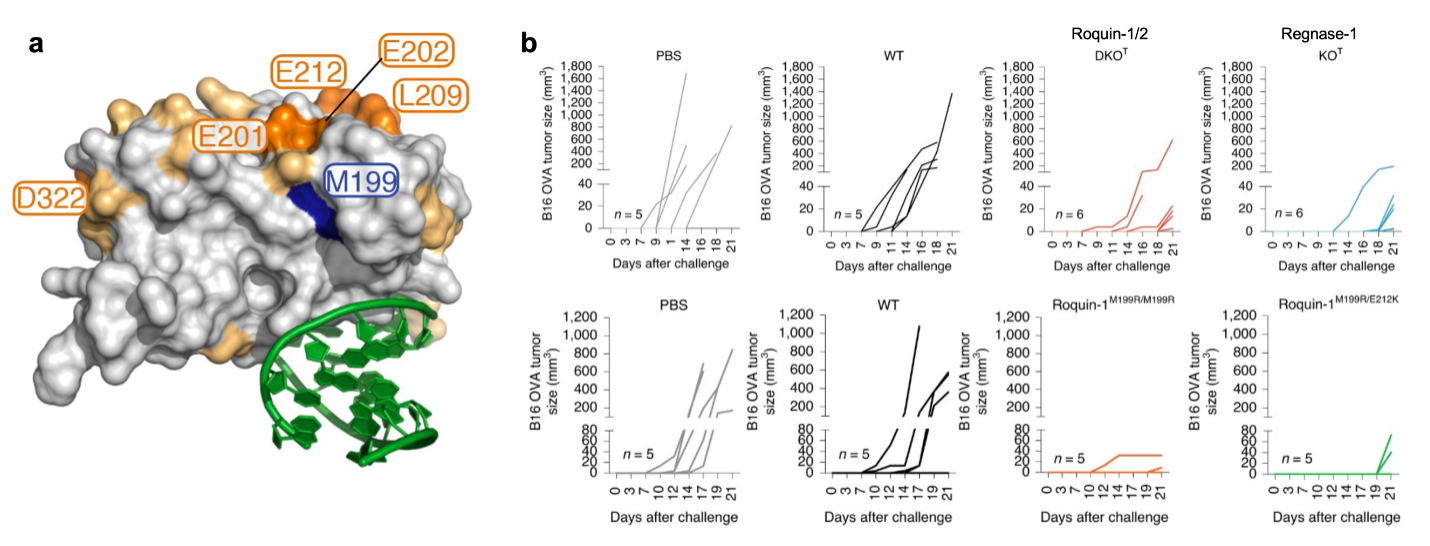
Regulators of the activation state of T cells are promising targets for immunotherapy. The research on Roquin-1 and Regnase-1 has revealed that these RNA-binding proteins prevent autoimmunity and are key regulators of proliferation, persistence, cytotoxicity and exhaustion in tumor-antigen-specific CD8+ T cells. In fact, individual loss-of-function of Regnase-1 or combined inactivation of Roquin-1 and Roquin-2 in T cells enabled superior control of tumor growth. Nevertheless, the inactivation of these factors also caused autoimmunity and neurotoxicity. In the current funding period, we have addressed how physiologic regulation of Roquin by the paracaspase MALT1 affects T cell responses.
We demonstrated that MALT1-induced autoimmunity only developed, if MALT1 was able to cleave Roquin-1. Conversely, antigen-specific CD8+ T cells, harboring mutations that render Roquin-1 and Roquin-2 insensitive to MALT1, were unable to mount antitumor responses. Our preliminary work has established that Roquin-1 and Regnase-1 form a ternary complex on RNA, and we identified specific residues in the ROQ domain of Roquin-1 that enable the interaction with Regnase-1. Mutations of these residues that weaken Roquin-1/Regnase-1 interaction and interfere with cooperative target regulation cause autoimmunity and enhance antitumor immunity. Based on these findings, we propose to develop innovative strategies to target Roquin and Regnase-1 function. Modulating Roquin/Regnase activities transiently, to avoid autoimmunity, we will define the molecular and cellular consequences for T cells within the tumor. Specifically, in Aim 1 we will employ de novo protein design to create binders for mouse and human Roquin proteins that are able to shield two interaction sites in the ROQ domain of Roquin and interfere with its binding to Regnase-1 or RNA. In Aim 2 we will evaluate the therapeutic benefit of expressing these artificial molecules in adoptively transferred antigen-specific T cells that are infiltrating tumors and control tumor growth. We will investigate the effects of constitutive or transient ectopic expression of the binder. In Aim 3, we will compare how acutely impaired Roquin/Regnase cooperation and impaired binding to RNA as well ubiquitination-dependent depletion of Roquin proteins impose specific molecular and cellular changes that strengthen antitumor immunity.
More