Title
Bench-to-Bedside ATMP Development Platform
Research Area
Good Manufacturing Practice (GMP) Good Clinical Practice (GCP)
Project Summary
Project Z02 serves as a structural backbone to foster clinical translation, risk awareness, ethical reflection and regulatory compliance in a structured manner. This is pivotal for the CRC as a whole, as projects learn and benefit from each other in the translational process and build upon a growing body of experience in the CRC while avoiding erroneous developments as early as possible.
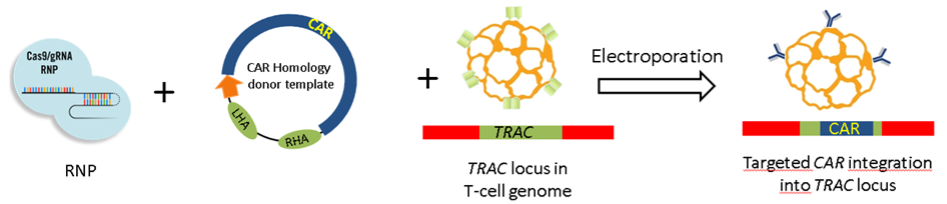
Within the CRC, several gene-engineered T cell products (advanced therapy medicinal products, ATMPs in ‘regulatory’ terminology) are being developed. The ambition is to advance at least 1-2 ATMPs per year into first-in-human (FIH) clinical trials, even though funding for such clinical trials will have to procured from other funding schemes (e.g. DZF, DKTK, BZKF, NCT, BMBF). Project Z02 serves as a gateway infrastructure and service project to enable and facilitate the clinical translation of selected ATMPs from the CRC portfolio.
Accordingly, the PIs of this project Z02 have been selected based on their distinguished expertise in clinical trial design and conduct in cellular therapy (Einsele), regulatory affairs and Good Manufacturing Practice (GMP, Hildebrandt / Lindemann), and ethical implications of individualized genomic treatment strategies (Marckmann).
As such, Z02 has grown into a catapult position, with a definition of an entry point, guidance, milestones and a “doable” target, i.e. the application for Early Entry Status at the European Medicine Agency’s PRIME scheme for further regulatory guidance. The first projects have been accompanied and guided within the CRC, making them a blueprint for next projects to follow the path defined by Z02.
An alignment of quality assurance structures in Würzburg and Munich has been fostered, and the specialized programs SOPGuard, an electronic QA documentation system, and FormGuard, for the management and recording of QM, were established and qualified.
The GMP infrastructure has grown substantially, providing projects with critical technology platforms needed for the manufacture of first clinical batches. Key vendors have been qualified for key processes in gene engineering in a clinical context.
A set of regulatory meetings, audits, technical, clinical and ethics workshops has helped to define the path towards clinical translation, the key responsibility of Z02 within the CRC.
Goals of the next funding period will include the submission of the first project(s) to the European Medicine Agency (EMA) for consideration of their inclusion in EMA’s PRIME scheme and a structured promotion of the internal and external discourse about the ethical implications of the CRC’s work.