Title
Engineering CAR-T cells against neuroblastoma with synthetic notch receptors to increase tumor-specificity and endogenous immune activation
Research Area
CAR-T cell therapy
Project Summary
Here we summarize the output and findings from Project A06, conducted within the first funding period of TRR338, and aiming to improve CAR-T cell therapies for neuroblastoma, a pediatric solid tumor with an immunologically cold tumor microenvironment. The research focused on engineering CAR-T cells with synthetic notch receptors to improve tumor specificity and persistence, as well as developing an allogeneic, “off-the-shelf” CAR-T cell product for faster and more accessible treatment. A06 also investigated the potential to arm CAR-T cells with cytokines to enhance their function within the tumor microenvironment and prevent tumor escape. In line with TRR338 goals to advance understanding in cancer immunotherapy, A06 aimed to create more effective and safer CAR-T cell therapies for solid tumors, using the test case, neuroblastoma.
Key Findings
Project A06 produced several key findings that contribute to advancing knowledge in the field driving preclinical development and testing of CAR-T cell therapies to treat neuroblastoma. These findings include:
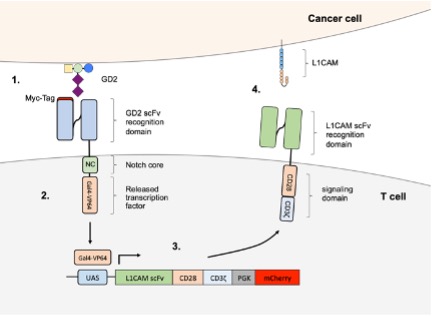
1. The use of SynNotch-driven gene expression with synNotch receptors targeting common neuroblastoma antigens is less effective compared to the well-established CD19-targeting synNotch receptor and requires significant refinement to ensure strong induction without unwanted leakage.
2. Primary T cells were effectively modified with multiple gene knockouts and equipped with various designs of HLA-E/B2M fusion proteins. However, HLA-E/B2M overexpression provided only partial protection against NK cells, prompting us to explore additional targets to reduce NK cell-mediated killing of MHC class I-deficient T cells.
3. The kinetically unstable IL12 variants developed by CRC partner A04 (M. Feige) showed comparable functionality to wildtype IL12, but without causing toxicity in an immunocompetent neuroblastoma mouse model, a major challenge in IL12-based immunotherapies. CAR-T cells expressing either wildtype or destabilized IL12 exhibited a less differentiated phenotype compared to CAR-T cells lacking the IL12 component. This phenotype remained stable even after multiple rounds of stimulation with antigen-expressing tumor cells, suggesting a potential benefit for the CAR-T cell persistence and long-term antitumor activity (manuscript in preparation).
4. Together with CRC partner M. Eilers, we revealed that a primary oncogenic driver in neuroblastoma, MYCN, downregulates L1CAM expression, which is a target for CAR-T cells (Grunewald et al, 2025).
Project-Related Publications
Künkele, A., Johnson, A.J., Rolczynski, L.S., Chang, C.A., Hoglund, V., Kelly-Spratt, K.S., Jensen, M.C et al: Functional Tuning of CARs Reveals Signaling Threshold above Which CD8+ CTL Antitumor Potency Is Attenu- ated due to Cell Fas-FasL-Dependent AICD. Cancer Immunol Res 2015; 3, 368-379
Künkele, A., Taraseviciute, A., Finn, L.S., Johnson, A.J., Berger, C., Finney, O., Chang, C.A., Rolczynski, L.S., Brown, C., Mgebroff, S., Berger, M., Park, J.R., Jensen, M.C.: Preclinical Assessment of CD171-Directed CAR T-cell Adoptive Therapy for Childhood Neuroblastoma: CE7 Epitope Target Safety and Product Manufacturing Feasibility. Clin Cancer Res 2017; 23, 466-477
Künkele, A., Brown, C., Beebe, A., Mgebroff, S., Johnson, A.J., Taraseviciute, A., Rolczynski, L.S., Chang, C.A., Finney, O., Park, J.R., Jensen, M.C.: Manufacture of chimeric antigen receptor T cells from mobilized cryo- preserved peripheral blood stem cell units depends on monocyte depletion. Biol Blood Marrow Transplant 2019; 25:223-232
Andersch, L., Radke, J., Klaus, A., Schwiebert, S., Winkler, A., Schumann, E., Grunewald, L., Zirngibl, F., Flem- mig, C., Jensen, M.C., Rossig, C., Joussen, A., Henssen, A., Eggert, A., Schulte, J.H., Künkele A: CD171- and GD2-specific CAR T cells potently target retinoblastoma cells in preclinical in vitro testing. BMC Cancer 2019; 19, 895
Ali, S., Toews, K., Schwiebert, S., Klaus, A., Winkler, A., Grunewald, L., Oevermann, L., Deubzer, H.E., Tüns, A., Jensen, M.C., Henssen, A.G., Eggert, A., Schulte, J.H., Schwich, E., Rebmann, V., Schramm, A., Künkele A: Tumor-derived extracellular vesicles impair CD171-specific CD4+ CAR T cell efficacy. Front Immunol 2020; 11:531
Toews, K., Grunewald, L., Schwiebert, S., Klaus, A., Winkler, A., Ali, S., Zirngibl, F., Astrahantseff, K., Wagner, D.L., Henssen, A.G., Deubzer, H.E., Schulte, J.H., Ochsenreither, S., Eggert, A., Künkele A: Central memory phenotype drives success of checkpoint inhibition in combination with CAR T cells. Mol Carcinog 2020; 59:p.724- 735