Title
Overcoming Barriers for T cell Therapy in the Liver for Hepatitis B and Hepatocellular Carcinoma (HCC)
Research Area
Virology, Immune Therapy, Virus-induced Cancer
Project Summary
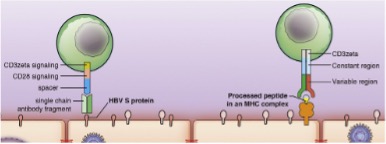
Over 250 million humans are chronically infected with the hepatitis B virus (HBV). HBV is controlled by natural T cell immunity in most adults. However, this control fails if the infection is acquired early in life and chronic infection develops, causing liver disease and HCC. The death toll of >1 million HBV carriers per year shows the high medical need for new treatment approaches. Our goal is to establish T cell therapy to eliminate hepatocytes persistently infected with HBV, particularly those that carry HBV integrations and serve as precursors of liver cancer. The HBV small envelope protein S is a promising target for T cell therapy because S is continuously expressed in persistent HBV infection, as well as when HBV-DNA has integrated into hepatocytes and in most HBV-related hepatocellular carcinoma. S-expression can be detected in blood by secreted HBsAg. The elimination of HBsAg indicates the elimination of HBV, a significantly reduced cancer risk, and an improved prognosis, and thus is the primary treatment goal (1). The first clinical applications of TCR-T cells engineered with our HBV S-specific TCRs in patients with late-stage HBV-associated HCC reduced or eliminated HBsAg after lymphodepletion. This initiated the discussion in the field on whether T cell therapy is a suitable approach to cure HBV infection.
The aim of this project is to optimize effector function, expansion, and survival of transferred HBV-specific TCR-T cells to improve their use in the setting of chronic HBV infection, where lymphodepletion may not be applied. Furthermore, we will exploit the role of CD4+ T cell help in preclinical models. We will decipher whether antigen recognition of MHC-II by the CD4+ T cells is required, and what role cytokines play in the helper function of CD4+ T cells. In addition, we will investigate the role of 4-1-BB co-activation and PD-1 checkpoint inhibition to improve the expansion, survival, and antiviral function of TCR-engineered T cells and use PD-1 immunoswitch receptors to convert co-inhibitory into costimulatory signals. This shall serve as the basis to develop clinical protocols to treat chronic hepatitis B by T cell therapy.